- Englist


The Ultimate Guide to Conductive Graphite Powder for Industrial Applications
In the realm of advanced materials, conductive graphite powder has emerged as a cornerstone for enhancing electrical conductivity in polymers, coatings, and lubricants. By leveraging the unique hexagonal crystal structure of graphite, manufacturers can transform insulating materials into functional conductive composites. Whether it is for EMI shielding, antistatic flooring, or high-performance battery electrodes, the right grade of graphite powder is essential for stability and efficiency. In this guide, we will explore the technical nuances and industrial benefits of using this versatile material to optimize your product's performance.
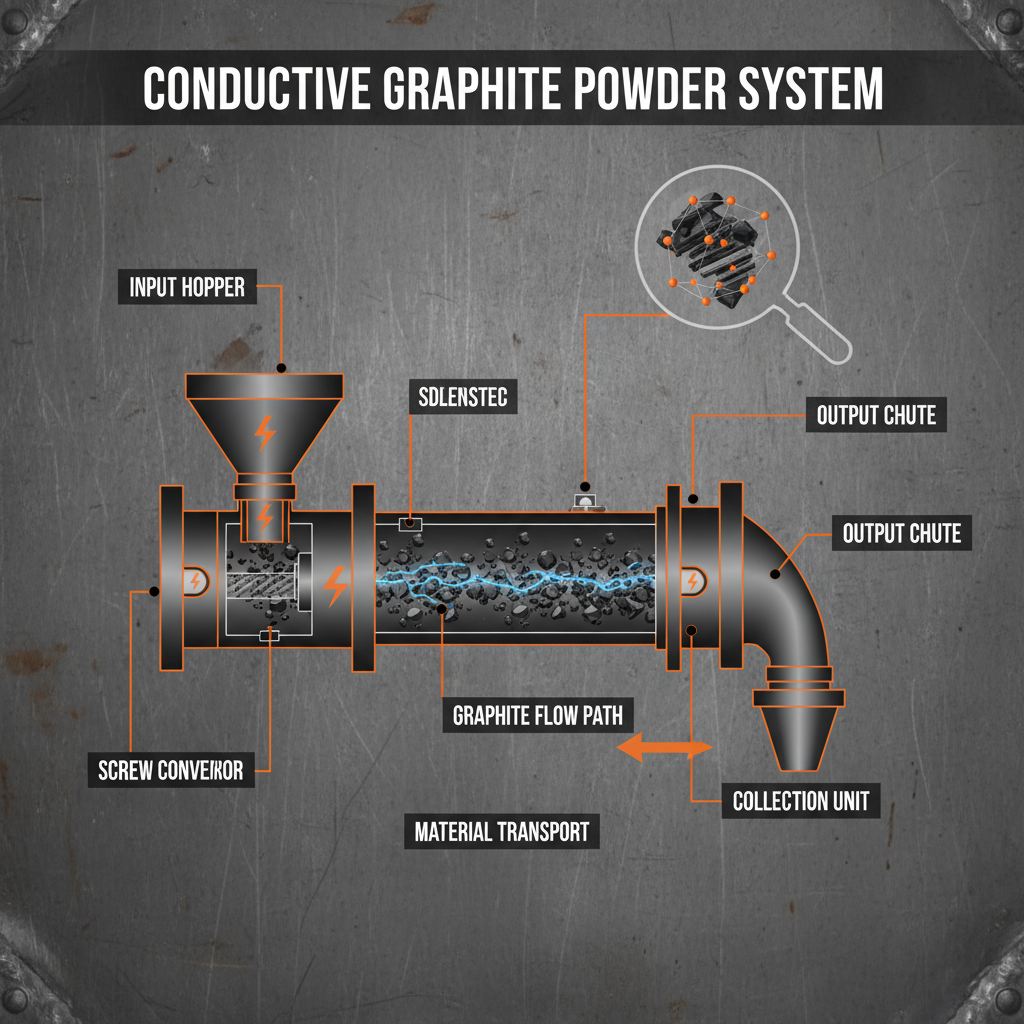
Understanding the Mechanism of Conductive Graphite Powder
The effectiveness of conductive graphite powder relies on the concept of percolation threshold. This is the critical concentration where the graphite particles form a continuous network throughout the matrix, allowing electrons to flow freely. Because graphite possesses high intrinsic conductivity and excellent thermal stability, it is often preferred over metal powders due to its lower density and superior corrosion resistance. The particle size and morphology—whether spherical, flaky, or irregular—play a vital role in determining how much powder is needed to achieve the desired conductivity level without compromising the mechanical integrity of the host material.
Pro Tip: To minimize the amount of filler required, choose a powder with a high aspect ratio (flaky graphite), as it creates a conductive network more efficiently than spherical particles.
Key Properties and Specifications of Conductive Graphite Powder
When sourcing conductive graphite powder, technical specifications are the primary indicators of performance. Purity is critical, as impurities can increase electrical resistance and affect the chemical stability of the composite. Most industrial-grade powders are categorized by their carbon content and average particle size (D50). For precision electronics, ultra-fine powders are used to ensure a smooth surface finish and uniform conductivity. Below is a detailed specification table showing common grades used in the industry.
Industrial Applications of Conductive Graphite Powder
The versatility of conductive graphite powder makes it indispensable across several high-tech sectors. In the automotive industry, it is used to create antistatic components that prevent spark discharge in fuel systems. In aerospace, graphite-filled polymers are utilized for electromagnetic interference (EMI) shielding to protect sensitive avionics from external noise. Additionally, the energy sector relies heavily on graphite for battery anodes and fuel cell plates, where electrical conductivity and chemical stability are non-negotiable requirements. By integrating these powders, engineers can achieve a precise balance between weight reduction and electrical performance.

Comparing Conductive Graphite Powder vs. Carbon Black
A common dilemma for engineers is choosing between conductive graphite powder and carbon black. While both are carbon-based, they offer very different properties. Graphite has a crystalline structure that provides significantly higher electrical conductivity per particle. Carbon black, on the other hand, is amorphous and is often used more for reinforcement and UV protection than for high-level conductivity. Graphite typically requires a lower loading percentage to reach the percolation threshold, which helps in maintaining the flexibility and impact strength of the final composite material.
Key Comparison Highlights:
• Conductivity: Graphite is superior for electrical transport.
• Loading: Graphite typically requires lower volume fractions.
• Lubrication: Graphite provides inherent self-lubricating properties.
• Structure: Crystalline (Graphite) vs. Amorphous (Carbon Black).
How to Optimize the Integration of Conductive Graphite Powder
To maximize the efficiency of conductive graphite powder, proper dispersion is key. Agglomeration of particles can lead to "dead zones" in the material, resulting in inconsistent conductivity and structural weak points. Utilizing high-shear mixing or ultrasonic dispersion techniques ensures that the graphite is evenly distributed throughout the polymer matrix. Furthermore, the use of coupling agents can improve the interfacial adhesion between the inorganic graphite and the organic resin, preventing delamination and improving the overall mechanical durability of the part.
Environmental and Safety Considerations for Graphite Powders
While conductive graphite powder is chemically inert and non-toxic, its physical form as a fine powder requires careful handling. Inhalation of fine dust can irritate the respiratory system, and graphite's conductive nature means that airborne powder can cause short circuits if it settles on open electronic equipment. Industrial facilities should employ dust extraction systems and ensure workers wear appropriate PPE, such as N95 masks. From an environmental perspective, graphite is a sustainable alternative to many heavy-metal-based conductive fillers, making it a preferred choice for companies striving for "green" manufacturing certifications.
Conclusion: Elevating Performance with Conductive Graphite Powder
The strategic use of conductive graphite powder allows manufacturers to bridge the gap between insulating plastics and conductive metals. By selecting the appropriate particle size, ensuring high purity, and optimizing dispersion, you can create high-performance materials that meet the rigorous demands of modern electronics and automotive engineering. As the industry moves toward smarter, lighter, and more efficient components, graphite continues to be an essential tool in the material scientist's arsenal. Invest in quality graphite solutions to ensure the longevity and reliability of your products.
Frequently Asked Questions (FAQs)
How does particle size affect the conductivity of graphite powder?
Particle size significantly impacts both the electrical properties and the processing of the material. Smaller, ultra-fine particles provide a higher surface area, which can lead to a more uniform distribution in a composite; however, they may require higher loading to reach the percolation threshold compared to larger, flaky particles. Flaky graphite particles act as "bridges" between other particles, creating a conductive path more quickly. Therefore, the choice depends on whether you prioritize surface smoothness (fine powder) or maximum conductivity with minimum filler (flaky powder).
Can conductive graphite powder be used in water-based coatings?
Yes, it can be used in water-based coatings, but it requires the addition of appropriate surfactants or dispersing agents. Because graphite is naturally hydrophobic (it repels water), it tends to clump or float on the surface of water-based mixtures. By using a wetting agent, the surface tension is reduced, allowing the conductive graphite powder to integrate seamlessly into the aqueous medium. This is commonly done for antistatic paints and conductive primers used in industrial flooring.
What is the difference between synthetic and natural graphite powder?
Natural graphite is mined and then purified; it generally offers a cost-effective solution with excellent flaky structures ideal for conductivity. Synthetic graphite is produced through the graphitization of petroleum coke or coal tar pitch at extremely high temperatures. Synthetic graphite often has higher purity levels and more consistent particle shapes, which is critical for high-end battery applications or specialized semiconductor components. While natural graphite is sufficient for most antistatic and EMI shielding tasks, synthetic is preferred where chemical purity is the top priority.
How do I store conductive graphite powder to prevent contamination?
To maintain the integrity of conductive graphite powder, it should be stored in a cool, dry environment in airtight containers. Because graphite is a lubricant and highly conductive, it can easily contaminate other materials in a workshop. Containers should be clearly labeled and kept away from strong oxidizing agents. Using moisture-proof bags or sealed drums prevents the powder from absorbing humidity, which can lead to clumping and negatively affect the precision of your weighing and mixing processes during production.





 Pervious
Pervious
 Next
Next




















